By Donald A. Wollheim, MD, WCC, DWC, FAPWCA
To begin appropriate treatment for chronic venous insufficiency (CVI), clinicians must be able to make the correct diagnosis. Part 1 (published in the March-April edition) described CVI and its presentation. This article provides details of the CVI diagnosis (including the differential diagnosis from other diseases), disease classification to help assess the extent of CVI, diagnostic studies used to diagnose CVI, and various treatment options to “rescue” the patient from CVI.
CVI diagnosis
CVI is diagnosed mainly from the patient’s history and physical exam. Typically with CVI, discomfort in the lower extremity gets worse toward the end of day, decreases with leg elevation, and feels the best first thing in the morning after the patient has slept with the legs elevated. Physical examination may reveal edema, acute or chronic leg changes, or both. Some extremities may show permanent changes, such as hemosiderin staining, atrophie blanche, or lipodermatosclerosis. Varicose veins also may be present. In general, the more severe CVI abnormalities usually occur in the older patients and those with significant comorbidities, such as peripheral arterial disease (PAD), diabetes mellitus, and arthritis. (For a glossary of terms, see Part 1 of this article in the March-April issue.)
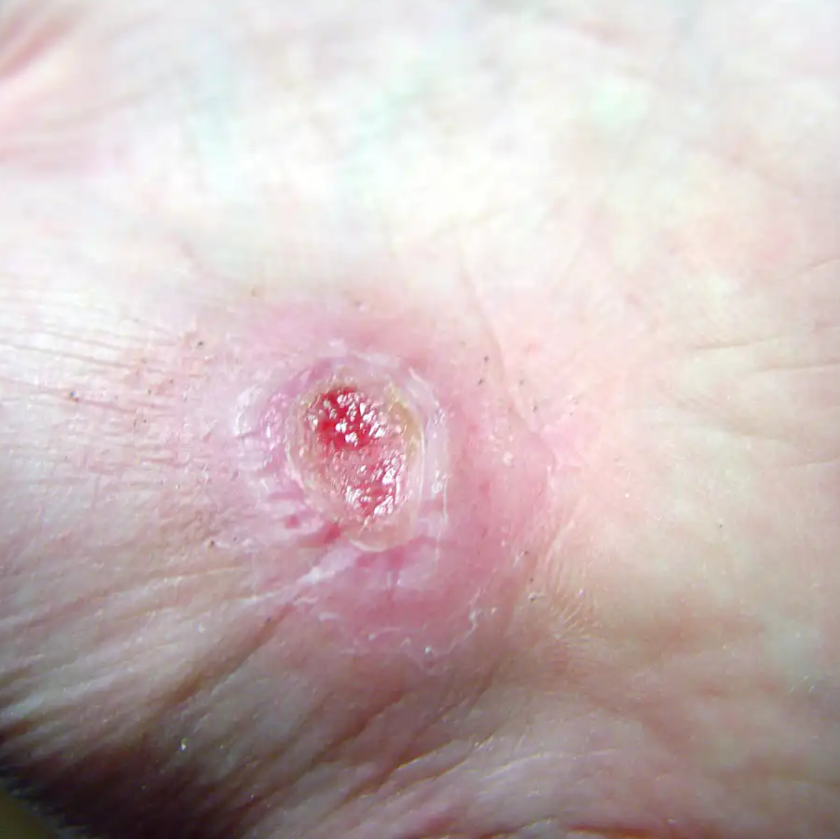
CVI ulcers, if present, commonly are superficial with a red wound bed. In keeping with the “broken water balloon” analogy described in Part 1, these ulcers may have a moderate amount of drainage. They tend to be located below the knee but not on the foot, and are more common on the medial aspect of the leg along the course of the greater saphenous vein. However, they may also be found on the lateral aspect of the lower leg along the course of the lesser saphenous vein.
Differential diagnosis
CVI must be differentiated from PAD and diabetic peripheral neuropathy to provide appropriate and safe therapy. However, a patient with CVI may also have PAD and/or diabetic peripheral neuropathy at the same time. Though the CVI changes usually occur above the foot, the changes for both peripheral arterial disease and diabetic neuropathy occur below the ankle.
PAD
In an extremity with PAD, the ankle-brachial index typically is 0.9 or lower. As noted above, some patients may have an extremity with both CVI and PAD. In this case, the therapy selected must accommodate both diseases, because PAD can hinder the healing of CVI wounds.
Pain associated with PAD differs from CVI-related pain. With CVI, pain improves with elevation, whereas the opposite may occur with PAD. Typically, PAD patients require gravity to aid arterial blood flow to the extremity and thus elevation of the PAD extremity may aggravate, not improve, the pain. PAD pain is linked to ischemia, is mild to severe, and stems from a more proximal (closer to the heart) vessel narrowing or occlusion.
PAD-related pain often is called claudication, an intermittent discomfort occurring with exercise, which may progress to nocturnal pain, discomfort caused by elevation of the nonexercising leg at night. It may progress to a more severe resting pain, where the pain is constantly present independent of the leg’s position or activity. Resting pain cannot be eliminated or significantly improved without some form of surgical intervention (which, unfortunately, may involve an amputation). Physical examination may show changes associated with decreased arterial blood flow, such as a lack of hair, diminished or absent pulses, and dystrophic (thick) toenails.
Also, arterial ulcers (from PAD) tend to be more painful than venous ulcers. They’re usually deeper than the more superficial venous ulcers, and are located at the distal part of the arterial vascular tree, near the toes or on the lateral aspect of the foot (the area more subject to footwear trauma). An arterial ulcer has a pale wound bed with decreased drainage due to impaired blood flow to the tissue. Because of the decreased blood flow, the ulcer may progress from reversible tissue ischemia to irreversible necrotic tissue. These ulcers commonly are also infected due to the compromised arterial blood flow.
Diabetic peripheral neuropathy
In a patient with diabetes, the leg may show a combination of the three types of neuropathy—peripheral motor, peripheral sensory, and peripheral autonomic neuropathy. Findings in these legs may differ significantly from a leg with CVI. However, like PAD, a CVI extremity may also have concurrent diabetic peripheral neuropathic changes.
In a patient with diabetic peripheral neuropathy, the history usually reveals at least a 10-year history of diabetes. Wounds are within an insensate (numb) foot and on its plantar aspect.
A diabetic foot ulcer typically starts as a callous on the bottom of the foot, which breaks down to create the ulcer. Commonly, a bony deformity is present due to neuropathy and acts as a focal point of increased shearing and pressure with walking. This leads to the initial callous formation. These wounds usually occur within the surrounding callous, in an insensate area of the plantar aspect of the foot, and commonly with an overlying bony foot deformity.
Disease classification
Several classification systems exist for CVI. One system that helps guide therapy is called CEAP.
CEAP: Clinical Etiology Anatomy Pathophysiology
CEAP classification should be done for all lower extremities with CVI. Therapy for CVI varies with signs and symptoms, disease extent and duration, and the overall treatment goal (be it that of symptomatic relief or cosmetic improvement). CEAP components include clinical severity, etiology, anatomy, and pathophysiology.
Clinical severity has eight grades:
• C0: no visible or palpable signs of venous insufficiency
• C1: telangiectasia, reticular veins, or both
• C2: varicose veins
• C3: edema into the skin and subcutaneous space, which may be pitting. (Note: Unlike lymphedema, CVI doesn’t involve the foot or toes.)
• C4a: pigmentary skin changes, such as hemosiderin staining or eczema
• C4b: lipodermatosclerosis
• C5: healed venous ulcer with or without atrophic skin; it may present as a pigmentary change, usually of lighter discoloration, within an area of hemosiderin staining
• C6: active venous ulcer.
Etiology may be classified as:
• Ep: primary
• Es: secondary (due to an etiology other than CVI, such as trauma)
• En: unknown (nonspecific)
• Ec: congenital.
Anatomy (location) of the pathologic vein may be classified as:
• As: superficial veins
• Ad: deep veins
• Ap: perforating veins
• An: nonspecific veins.
Pathophysiology may be classified as:
• Pr: Venous reflux due to valve damage or prior DVT leading to retrograde blood flow
• Po: Venous obstruction due to ongoing venous thrombosis
• Po, r: Both reflux and obstruction
• Pn: Neither reflux nor obstruction
Other classification systems
Other systems for classifying CVI include the Venous Clinical Severity Scale and Venous Disability Scale. (See Other CVI classification systems.)
Diagnostic studies
Ultrasonography, a noninvasive study, generally is less expensive than venography (the gold standard study for CVI) and carries no risk. In fact, duplex ultrasonography has largely replaced invasive venography. (See Rarely used studies.)
Ultrasonography indications include:
• diagnosis of venous obstruction, venous reflux, and deep vein thrombosis
• aid in the diagnosis of suspected CVI when that diagnosis can’t be established from signs and symptoms alone
• evaluation of patients with physical findings of CVI but with atypical symptoms
• evaluation of patients with atypical venous insufficiency, such as those younger than age 40
• evaluation of patients who develop CVI after trauma
• evaluation of patients with clinical CVI who don’t respond to standard therapy
• to rule out pathologic conditions that may mimic CVI
• to guide interventions by evaluating the anatomic and physiologic features of CVI.
Venography
Venography is considered the gold standard for diagnosing venous insufficiency, but is rarely needed due to the ultrasonographic techniques available today. During venography, contrast dye is injected into the patient’s venous system to identify the normal and abnormal anatomy of the blood flow within and out of the extremity.
Venography is more expensive than ultrasound studies. Unlike ultrasound, it’s invasive and can cause complications such as venous phlebitis, thrombosis, and reactions to the contrast dye.
Patients with lower-extremity wounds should undergo an arterial assessment to identify if the wound has an arterial component where there is not enough oxygen-rich blood flow to allow healing. This arterial evaluation is also necessary if compression therapy is ordered, which often is for the CVI extremity, to guide the clinician on how it can safely be applied.
Ankle-brachial index
Ankle-brachial index (ABI) is a noninvasive study that can diagnose PAD and determine how much lower-extremity compression can be applied safely in patients with CVI. ABI compares systolic blood pressure measured at the ankle with the systolic blood pressure measured at the arm. Normally, the pressure at the elbow (brachial artery) is the same as the pressure in the ankle arteries (dorsalis pedis or posterior tibial arteries).
The results of the ABI study should be used as a guide for ordering a safe level of compression in the CVI patient. If the ABI is not determined, the patient might develop significant complications from an incorrect amount of compression applied, leading to “downstream” ischemia and possibly necrosis of tissue.
A normal ABI result is 1. Anything below 1.0 indicates PAD. ABI of 1.3 or higher typically is a falsely elevated level as a result of an inability to compress calcified ankle vessels. This commonly occurs in elderly patients and those with diabetes or renal failure. An ABI of 1.3 or higher warrants another study to evaluate arterial blood flow to that extremity. (See Interpreting and applying ABI results in CVI patients by clicking the PDF icon above.)
Management guidelines
During the first 6 months after the diagnosis is made, conservative therapy should be used for CVI patients in an attempt to decrease the extremity swelling and reduce the “water balloon” effect—pooling of blood and intravascular fluid around the ankle (described in Part 1 of this article). Ways to do this include elevating the leg, initiating an exercise program, and using safe compression therapy. This conservative approach increases oxygen to the tissues, decreases edema (improving subcutaneous swelling, capillary constriction, and capillary separation), reduces tissue inflammation, and compresses the dilated veins.
If the patient has changes consistent with dermatitis or infection, use an appropriate topical agent—but be aware that this might preclude the use of long-wearing compression wraps to allow daily (or more frequent) access to the extremity for various medication applications.
If the patient has a noninfected ulcer, consider using a prolonged wearing wrap to provide compression, such as a long-stretch system (for example, multilayered wraps, single-layered wraps, or the Duke boot). Consider providing a resistance to leg swelling for ambulatory patients by using a short-stretch system, such as the Unna boot. Other options for these patients include adjustable compression wraps and intermittent pneumatic compression devices.
If the patient has an infected ulcer of the CVI extremity, consider using leg elevation plus a topical antimicrobial (with or without systemic antibiotic therapy), and postpone compression therapy until the infection is under control.
If the patient has no ulcers or a healed ulcer (through the proliferative phase of full-thickness wound healing), use removable compression stockings or an intermittent pneumatic pump system.
Ablative therapy
Unfortunately, the patient will need lifelong compression therapy if CVI can’t be corrected effectively with other more permanent forms of therapy. Clinicians might consider ablative therapy if symptoms do not improve after 6 months of conservative therapy or if the patient seeks cosmetic improvement. Ablative therapy refers to the various types of therapy that destroy or remove the abnormal veins.
Types of ablative therapy include chemical, thermal, and mechanical techniques.
• Chemical ablation consists of placing an irritating fluid directly into the vein, which damages the endothelial lining and leads to the destruction of the abnormality. This method is used mainly for telangiectasia, reticular veins, and small varicose veins. To treat larger veins without having to use large amounts of fluid, an ablative foam solution can be used instead; this may be done for larger varicosities, incompetent saphenous veins, or incompetent perforating veins.
• Thermal ablation uses a form of heat energy that destroys the wall of the vein. It may be used with topical light therapy applied to the skin for small, superficial dilated veins (telangiectasia or reticular veins). For larger veins (greater or lesser saphenous veins), heat energy may be applied within the vein itself using laser or radiofrequency energy.
• Mechanical ablation physically destroys or removes the abnormal vein. Examples of this technique include surgical vein ligation and vein stripping. (See Ablation success rates by clicking the PDF icon above.)
Correct identification is crucial
In a patient with suspected CVI, wound care clinicians must be able to correctly identify the disease process and determine if significant comorbid diseases are present before prescribing or administering any form of therapy. Otherwise, the therapy ordered may not correct the problem—and could even cause harm. Various diagnostic tools are available; in practice, ultrasonography is most often used for diagnosing venous disease.
Multiple therapeutic modalities are available to treat CVI. Unless the disease is corrected, the patient will need lifelong compression therapy. How much compression that can be safely applied depends on the presence and extent of comorbidities, such as PAD and heart failure.
Selected references
Alguire PC, Mathes BM. Clinical evaluation of lower extremity chronic venous disease. UpToDate. Last updated April 18, 2012. www.uptodate.com/contents/
clinical-evaluation-of-lower-extremity-chronic-venous-disease. Accessed April 11, 2013.
Alguire PC, Mathes BM. Diagnostic evaluation of chronic venous insufficiency. UpToDate. Last updated May 7, 2102. www.uptodate.com/contents/diagnostic-evaluation-of-chronic-venous-insufficiency?source=search_result&search=Diagnostic+evaluation
+of+chronic+venous+insufficiency&selectedTitle=1%7E128. Accessed April 11, 2013.
Alguire PC, Scovell S. Overview and management of lower extremity chronic venous disease. UpToDate. Last updated June 27, 2012. http://www.uptodate.com/
contents/overview-and-management-of-lower-extremity-chronic-venous-disease?source=search_result&search=Overview+and+management+of+lower+extremity+chronic+venous+disease&selectedTitle=1%7E150. Accessed April 11, 2013.
Moneta G. Classification of lower extremity chronic venous disorders. UpToDate. Last updated October 22, 2012. www.uptodate.com/contents/classification-of-lower-extremity-chronic-venous-disorders. Accessed April 11, 2013.
Sardina D. Skin and Wound Management Course; Seminar Workbook. Wound Care Education Institute: 2011;92-112.
Donald A. Wollheim is a practicing wound care physician in southeastern Wisconsin. He also is an instructor for Wound Care Education Institute and Madison College. He serves on the Editorial Board for Wound Care Advisor.



I have diagnost VI in lowere ankle and feet as well as P. neuropathy. I am not diabetic. Has any cause and effect been found between the 2 conditions? They both seemed to appear together